Diva Expertise
CRO/CDMO
Created in 2015
6-10 employees

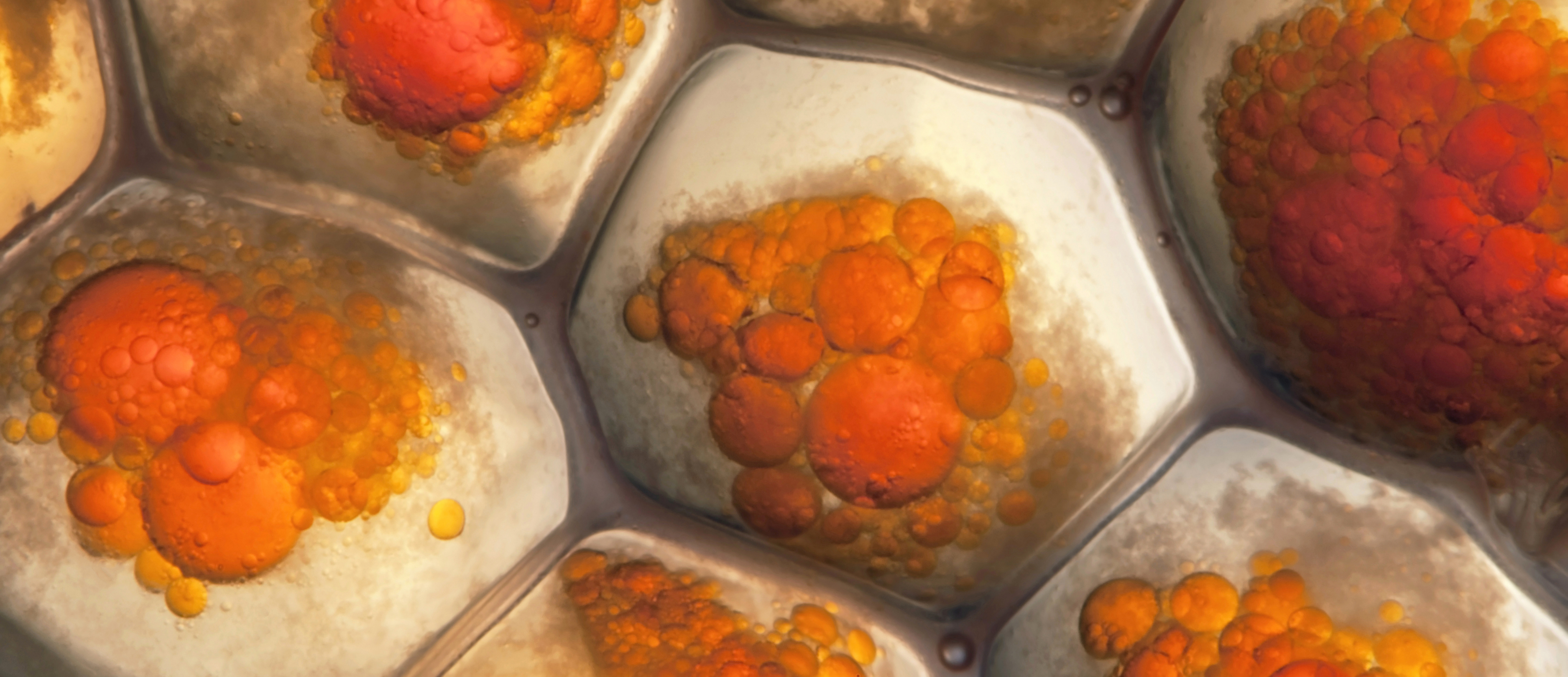
The expert of studies and supply in human adipose tissue
DIVA Expertise is the leading global scientific partner for companies in the health, nutraceuticals and cosmetics industries seeking to evaluate the efficacy of an active ingredient or product directly on human adipose and skin tissue, ex vivo and in vitro. DIVA Expertise offers turnkey R&D projects – using unique, scientifically validated predictive human biological models – to provide industrial players with robust mechanistic evidence that accelerates and ensures the safe market launch of their innovations in cosmetics, nutraceuticals, and pharma metabolism/obesity.
What sets DIVA apart: DIVA biobank offering and innovative in vitro and ex vivo models enable preclinical expertise that closely aligns with the physiology of human adipose tissue, by working with real human biological materials from donors and adhering to the 3Rs principle (reduction of animal testing).
CONTACT INFORMATION
Susan Benjamin
Business Developer
Toulouse, France
susan.benjamin@diva-expertise.com

DISCOVER MORE





































